In recent years, technological advancements have made it possible to create synthetic diamonds that have similar physical and chemical properties to natural diamonds. While synthetic diamonds are not considered “fake” or “imitation,” they are often more affordable than their natural counterparts, making them a popular choice for those who want the beauty of a diamond without the high cost. Synthetic diamonds are also often more environmentally friendly, as they do not require the same level of mining and extraction as natural diamonds.
In its pristine state, diamond is a non-conductive material, devoid of free electrons or “holes” that can facilitate electrical conduction (Figure 1). However, by introducing boron atoms into the diamond crystal lattice, its optical and electrical properties can be significantly altered. As the concentration of boron is increased, the diamond’s color shifts from its characteristic clear hue to a delicate shade of blue, while its electrical conductivity transforms from an insulator to a semiconductor.
Further increases in the boron content result in a lustrous blue shade that resembles the sheen of metallic surfaces and eventually culminates in a deep, ebony coloration. Such heavily boron-doped diamond (BDD) is also as electrically conducting as some metals, and at low temperatures, exhibits superconductivity, allowing electrical conduction with no resistance.
Superconducting diamond has drawn great interest due to its similarity to high-temperature superconductors, i.e., they are all doped insulators. Intuitively, in contrast to doped insulators, one would expect a good conductor to be more promising in establishing superconductivity, while the best conductors, i.e., gold and silver, do not behave as superconductors at all.
Two electrons that are bonded together and move as a single unit, known as Cooper pairs, must interact with and pass through numerous grain boundaries within the BDD film. Each grain boundary therefore acts like a weak link in the circuit. This gives rise to a series of exotic quantum phenomena, e.g., anomalous superconducting anisotropy and grain-sized dependent electrical transport.
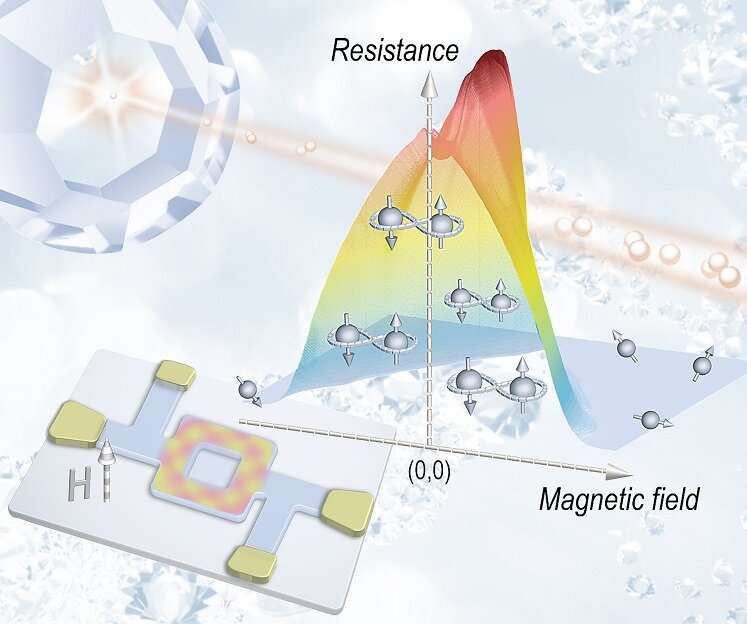
Figure 2. Unconventional giant “magnetoresistance” in diamond nanorings. © Gufei Zhang
Superconductivity or unconventional giant ‘magnetoresistance’ in diamond
Studies of superconductivity in various materials have established that in order for a material to become superconducting, it first needs to pass through a metallic state. A longstanding question is whether the formation of Cooper pairs in an established metal will inevitably transform the host material into a superconductor.
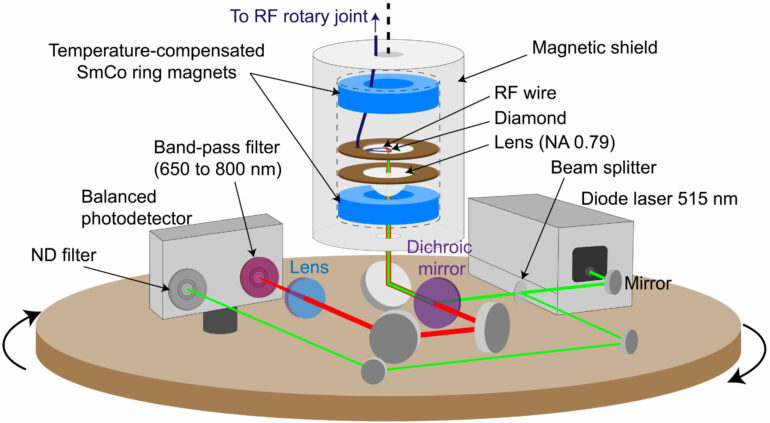
An international team of scientists led by researchers from Denmark, the U.K., China, Japan and Belgium has recently addressed this question by investigating the electrical transport in diamond nanorings—loops of superconducting BDD (Figure 2).
In their study published in Advanced Materials, they found that below the BDD superconducting transition temperature, the diamond nanorings demonstrate an abrupt resistance increase rather than a resistance drop, suggesting the transformation of the diamond nanorings from metals into bosonic semiconductors.
Along with the bosonic semiconducting transition, they observed a giant negative magnetoresistance that closely resembles the well-known giant magnetoresistance (GMR) in magnetic multilayers. In contrast to the conventional GMR originating from spin-dependent scattering, this unconventional giant negative “magnetoresistance” arises from the formation and trapping of Cooper pairs within the diamond nanorings. Demonstration of such exotic quantum phenomena may open up new paradigms for the development of novel superconducting quantum devices.
This story is part of Science X Dialog, where researchers can report findings from their published research articles. Visit this page for information about ScienceX Dialog and how to participate.
More information:
Gufei Zhang et al, Unconventional giant “magnetoresistance” in bosonic semiconducting diamond nanorings, Advanced Materials (2023). DOI: 10.1002/adma.202211129
Ramiz Zulkharnay recently started a new position as a Postdoctoral Research Associate at the University of Bristol. His research builds upon the foundation of his recently completed PhD thesis, entitled “Electron Emission Studies of Scandium on Diamond for Thermionic Solar Energy Generation Devices,” focusing on the surface functionalization of diamond for thermionic emission applications. With a background in Diamond for Energy, Ramiz has published several papers on the topic and presented his work at international conferences.
Citation:
Nanoscale ‘diamond rings’ provide unconventional giant ‘magnetoresistance’ for the development of new quantum devices (2023, March 9)