The 2024 Nobel Prize in chemistry recognized Demis Hassabis, John Jumper and David Baker for using machine learning to tackle one of biology’s biggest challenges: predicting the 3D shape of proteins and designing them from scratch.
This year’s award stood out because it honored research that originated at a tech company: DeepMind, an AI research startup that was acquired by Google in 2014. Most previous chemistry Nobel Prizes have gone to researchers in academia. Many laureates went on to form startup companies to further expand and commercialize their groundbreaking work – for instance, CRISPR gene-editing technology and quantum dots – but the research, from start to end, wasn’t done in the commercial sphere.
Although the Nobel Prizes in physics and chemistry are awarded separately, there is a fascinating connection between the winning research in those fields in 2024. The physics award went to two computer scientists who laid the foundations for machine learning, while the chemistry laureates were rewarded for their use of machine learning to tackle one of biology’s biggest mysteries: how proteins fold.
The 2024 Nobel Prizes underscore both the importance of this kind of artificial intelligence and how science today often crosses traditional boundaries, blending different fields to achieve groundbreaking results.
The challenge of protein folding
Proteins are the molecular machines of life. They make up a significant portion of our bodies, including muscles, enzymes, hormones, blood, hair and cartilage.
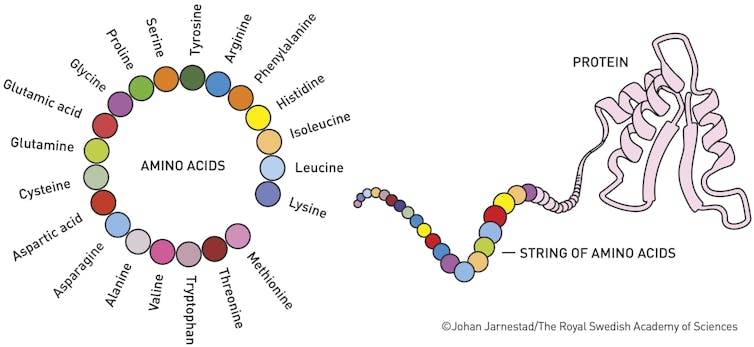
Proteins are chains of amino acid molecules that form a 3D shape based on their atoms’ interactions.
Johan Jarnestad/The Royal Swedish Academy of Sciences
Understanding proteins’ structures is essential because their shapes determine their functions. Back in 1972, Christian Anfinsen won the Nobel Prize in chemistry for showing that the sequence of a protein’s amino acid building blocks dictates the protein’s shape, which, in turn, influences its function. If a protein folds incorrectly, it may not work properly and could lead to diseases such as Alzheimer’s, cystic fibrosis or diabetes.
A protein’s overall shape depends on the tiny interactions, the attractions and repulsions, between all the atoms in the amino acids its made of. Some want to be together, some don’t. The protein twists and folds itself into a final shape based on many thousands of these chemical interactions.
For decades, one of biology’s greatest challenges was predicting a protein’s shape based solely on its amino acid sequence. Although researchers can now predict the shape, we still don’t understand how the proteins maneuver into their specific shapes and minimize the repulsions of all the interatomic interactions in a few microseconds.
To understand how proteins work and to prevent misfolding, scientists needed a way to predict the way proteins fold, but solving this puzzle was no…



