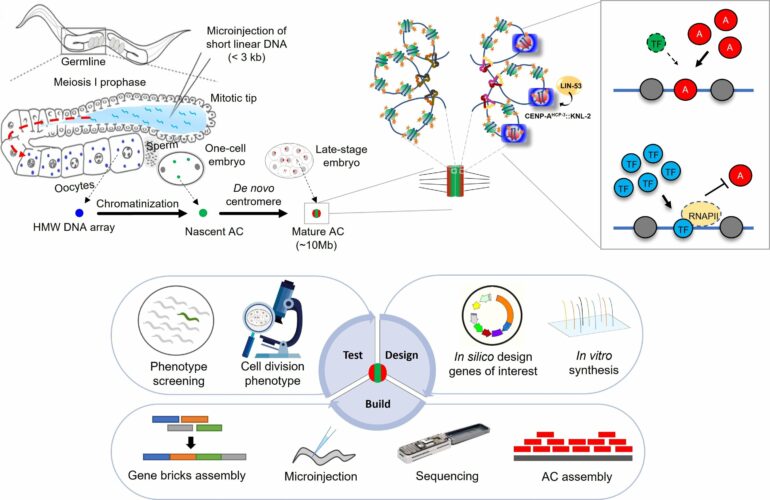
Human artificial chromosomes (HACs) capable of working within human cells could power advanced gene therapies, including those addressing some cancers, along with many laboratory applications, though serious technical obstacles have hindered their development. Now a team led by researchers at the Perelman School of Medicine at the University of Pennsylvania has made a significant breakthrough in this field that effectively bypasses a common stumbling block.
In a study published in Science, the researchers explained how they devised an efficient technique for making HACs from single, long constructs of designer DNA. Prior methods for making HACs have been limited by the fact that the DNA constructs used to make them tend to join together—”multimerize”—in unpredictably long series and with unpredictable rearrangements.
The new method allows HACs to be crafted more quickly and precisely, which in turn will directly speed up the rate at which DNA research can be done. In time, with an effective delivery system, this technique could lead to better engineered cell therapies for diseases like cancer.
“Essentially, we did a complete overhaul of the old approach to HAC design and delivery,” said Ben Black, Ph.D., the Eldridge Reeves Johnson Foundation Professor of Biochemistry and Biophysics at Penn. “The HAC we built is very attractive for eventual deployment in biotechnology applications, for instance, where large scale genetic engineering of cells is desired. A bonus is that they exist alongside natural chromosomes without having to alter the natural chromosomes in the cell.”
The first HACs were developed 25 years ago, and artificial chromosome technology is already well advanced for the smaller, simpler chromosomes of lower organisms such as bacteria and yeast. Human chromosomes are another matter, due largely to their greater sizes and more complex centromeres, the central region where X-shaped chromosomes’ arms are joined.
Researchers have been able to get small artificial human chromosomes to form from self-linking lengths of DNA added to cells, but these lengths of DNA multimerize with unpredictable organizations and copy numbers—complicating their therapeutic or scientific use—and the resulting HACs sometimes even end up incorporating bits of natural chromosomes from their host cells, making edits to them unreliable.
In their study, the Penn Medicine researchers devised improved HACs with multiple innovations: These included larger initial DNA constructs containing larger and more complex centromeres, which allow HACs to form from single copies of these constructs. For delivery to cells, they used a yeast-cell-based delivery system capable of carrying larger cargoes.
“Instead of trying to inhibit multimerization, for example, we just bypassed the problem by increasing the size of the input DNA construct so that it naturally tended to remain in predictable single-copy form,” said Black.
The researchers showed that their method was much more efficient at forming viable HACs compared to standard methods, and yielded HACs that could reproduce themselves during cell division.
The potential advantages of artificial chromosomes—assuming they can be delivered easily to cells and operate like natural chromosomes—are many. They would offer safer, more productive, and more durable platforms for expressing therapeutic genes, in contrast to virus-based gene-delivery systems which can trigger immune reactions and involve harmful viral insertion into natural chromosomes.
Normal gene expression in cells also requires many local and distant regulatory factors, which are virtually impossible to reproduce artificially outside of a chromosome-like context. Moreover, artificial chromosomes, unlike relatively cramped viral vectors, would permit the expression of large, cooperative ensembles of genes, for example to construct complex protein machines.
Black expects that the same broad approach his group took in this study will be useful in making artificial chromosomes for other higher organisms, including plants for agricultural applications such as pest-resistant, high-yield crops.
Researchers from the J. Craig Venter Institute, the University of Edinburgh, and the Technical University Darmstadt were also involved in the study.
More information:
Craig W. Gambogi et al, Efficient formation of single-copy human artificial chromosomes, Science (2024). DOI: 10.1126/science.adj3566
Provided by
Perelman School of Medicine at the University of Pennsylvania
Citation:
Scientists create novel technique to form human artificial chromosomes (2024, March 21)