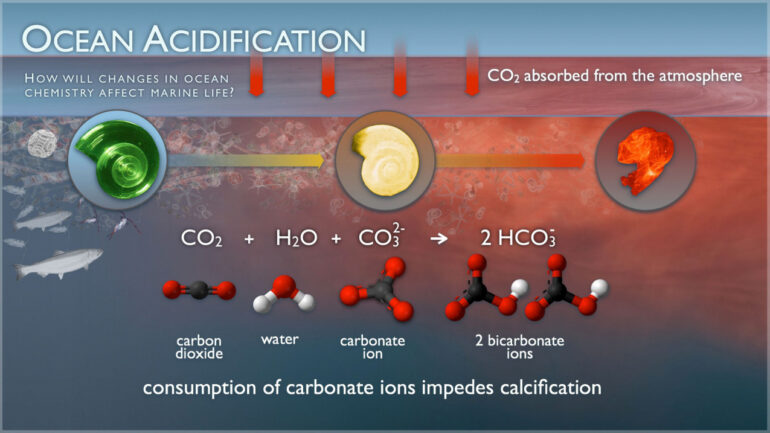
A new study demonstrates the important role of a common group of marine calcifying phytoplankton (coccolithophores) in the regulation of carbon dioxide (CO2) concentrations in the atmosphere.
The ocean has removed roughly a third of the CO2 released by humans since the Industrial Revolution. It is one of the largest sinks of anthropogenic CO2 and the largest reservoir of carbon that can easily exchange with the atmosphere on our planet. Understanding the processes that control the exchange of carbon between the ocean and atmosphere is key for projecting the future effects of carbon dioxide on climate change, ocean acidification, marine organisms, and society.
Research led by the Institute of Environmental Science and Technology of the Universitat Autònoma de Barcelona (ICTA-UAB) in collaboration with an international scientific team discovered that the exchange of carbon between the atmosphere and ocean is highly modulated by a unique group of photosynthesizing plankton called coccolithophores.
These common microscopic organisms which live in the sun-lit layer of the world’s oceans form elaborate plates of the carbon bearing mineral calcium carbonate. These layers are visible, for example, in places like the White Cliffs of Dover.
In the study published today in the journal Nature Communications, the team co-led by ICREA research professor Dr. Patrizia Ziveri at the ICTA-UAB found that coccolithophores dominate the production of CaCO3.
Coccolithophores are tiny algae which measure less than one hundredth of a millimeter and form the basis of the aquatic food web and contribute to the regulation of atmospheric CO2 levels through calcification and photosynthesis.
The study shows coccolithophores comprise 90% of the total production of CaCO3 in the surface ocean, indicating they play a key role in controlling ocean chemistry and CO2. This research highlights that the other two main planktonic calcifying groups, zooplankton (pteropods) and foraminifera, play a secondary role in the context of atmospheric CO2 modulation.
The study also revealed that rather than sinking into the deep ocean, a large portion of this CaCO3 dissolves close to the surface where carbon is more readily exchanged with the atmosphere and where sunlight penetrates the surface (photic zone).
“This extensive shallow dissolution explains the apparent discrepancy between previous estimates of CaCO3 production derived from satellite observations/biogeochemical modeling versus sinking particle estimates from shallow sediment traps,” explains Ziveri, who clarifies that the finding suggests that the processes driving shallow CaCO3 dissolution are key to understanding the role of planktonic calcifiers in regulating atmospheric CO2. This is important as more dissolution will increase the ability of water to hold CO2.
Dr. William Gray, a research scientist at the Laboratoire des Sciences du Climat et de l’Environnement who co-led the study, explains, “The dissolution of so much CaCO3 close to the ocean’s surface shows the exchange of carbon between the ocean and atmosphere is much more complicated than we first thought. Until we better understand the processes driving this shallow dissolution, it will be difficult to predict how the ocean will uptake carbon in the future.”
The study, which also involved ICTA-UAB researchers Michael Grelaud, Sven Pallacks and Griselda Anglada-Ortiz, used data obtained during a 2017 expedition to the North Pacific Ocean from Hawaii to Alaska and was produced in collaboration with an international scientific team with institutions from five countries.
More information:
Patrizia Ziveri et al, Pelagic calcium carbonate production and shallow dissolution in the North Pacific Ocean, Nature Communications (2023). DOI: 10.1038/s41467-023-36177-w
Provided by
Autonomous University of Barcelona
Citation:
New finding provides better understanding of oceans’ capacity to absorb atmospheric carbon dioxide (2023, February 20)