The 2023 Nobel Prize for chemistry isn’t the first Nobel awarded for research in nanotechnology. But it is perhaps the most colorful application of the technology to be associated with the accolade.
This year’s prize recognizes Moungi Bawendi, Louis Brus and Alexei Ekimov for the discovery and development of quantum dots. For many years, these precisely constructed nanometer-sized particles – just a few hundred thousandths the width of a human hair in diameter – were the darlings of nanotechnology pitches and presentations. As a researcher and adviser on nanotechnology, I’ve even used them myself when talking with developers, policymakers, advocacy groups and others about the promise and perils of the technology.
The origins of nanotechnology predate Bawendi, Brus and Ekimov’s work on quantum dots – the physicist Richard Feynman speculated on what could be possible through nanoscale engineering as early as 1959, and engineers like Erik Drexler were speculating about the possibilities of atomically precise manufacturing in the the 1980s. However, this year’s trio of Nobel laureates were part of the earliest wave of modern nanotechnology where researchers began putting breakthroughs in material science to practical use.
Quantum dots brilliantly fluoresce: They absorb one color of light and reemit it nearly instantaneously as another color. A vial of quantum dots, when illuminated with broad spectrum light, shines with a single vivid color. What makes them special, though, is that their color is determined by how large or small they are. Make them small and you get an intense blue. Make them larger, though still nanoscale, and the color shifts to red.
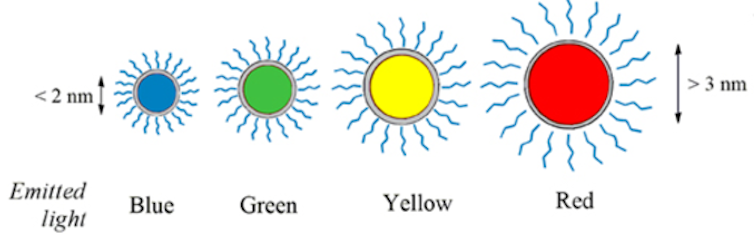
The wavelength of light a quantum dot emits depends on its size.
Maysinger, Ji, Hutter, Cooper, CC BY
This property has led to many arresting images of rows of vials containing quantum dots of different sizes going from a striking blue on one end, through greens and oranges, to a vibrant red at the other. So eye-catching is this demonstration of the power of nanotechnology that, in the early 2000s, quantum dots became iconic of the strangeness and novelty of nanotechnology.
But, of course, quantum dots are more than a visually attractive parlor trick. They demonstrate that unique, controllable and useful interactions between matter and light can be achieved through engineering the physical form of matter – modifying the size, shape and structure of objects or instance – rather than playing with the chemical bonds between atoms and molecules. The distinction is an important one, and it’s at the heart of modern nanotechnology.
Skip chemical bonds, rely on quantum physics
The wavelengths of light that a material absorbs, reflects or emits are usually determined by the chemical bonds that bind its constituent atoms together. Play with the chemistry of a material and it’s possible to fine-tune these bonds so that they give you…



