A team from the University of Chicago has announced the first evidence for “quantum superchemistry”—a phenomenon where particles in the same quantum state undergo collective accelerated reactions. The effect had been predicted, but never observed in the laboratory.
The findings, published July 24 in Nature Physics, open the door to a new field. Scientists are intensely interested in what are known as “quantum-enhanced” chemical reactions, which could have applications in quantum chemistry, quantum computing, and other technologies, as well as in better understanding the laws of the universe.
“What we saw lined up with the theoretical predictions,” said Cheng Chin, a professor of physics and member of the James Franck Institute and Enrico Fermi Institute, whose lab conducted the research. “This has been a scientific goal for 20 years, so it’s a very exciting era.”
Bose enhancement
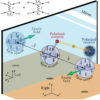
Chin’s lab specializes in working with particles held at very, very low temperatures. Near absolute zero, particles can link up so that they are all in the same quantum state—where they can display unusual abilities and behaviors.
It had been theorized that a group of atoms and molecules in the same quantum state would behave differently during chemical reactions, but the difficulty in orchestrating the experiment meant it had never been observed.
Chin’s group is experienced with herding atoms into quantum states, but molecules are larger and much more complex than atoms—so the group had to invent new techniques to wrangle them.
In the experiments, the scientists cooled down cesium atoms and coaxed them into the same quantum state. Next, they watched as the atoms reacted to form molecules.
In ordinary chemistry, the individual atoms would collide, and there’s a probability for each collision to form a molecule. However, quantum mechanics predicts that atoms in a quantum state perform actions collectively instead.
“You are no longer treating a chemical reaction as a collision between independent particles, but as a collective process,” explained Chin. “All of them are reacting together, as a whole.”
One consequence is that the reaction happens faster than it would under ordinary conditions. In fact, the more atoms in the system, the faster the reaction happens.
Another consequence is that the final molecules share the same molecular state. Chin explained that the same molecules in different states can have different physical and chemical properties—but there are times when you want to create a batch of molecules in a specific state. In traditional chemistry, you’re rolling the dice. “But with this technique, you can steer the molecules into an identical state,” he said.
Shu Nagata, a graduate student and co-author on the paper, added that they saw evidence that the reaction was taking place as a three-body interaction more often than as a two-body interaction. That is, three atoms would collide; two would form a molecule, and the third remained single. But the third played some role in the reaction.
Technological possibilities
The scientists hope the breakthrough is the start of a new era. Though this experiment was performed with simple, two-atom molecules, they plan to work their way up to handling larger and more complex molecules.
“How far we can push our understanding and our knowledge of quantum engineering, into more complicated molecules, is a major research direction in this scientific community,” said Chin.
Some in the field have envisioned using molecules as qubits in quantum computers, or in quantum information processing, for example. Other scientists are exploring them as gateways to even more precise measurements of fundamental laws and interactions, such as testing basic laws of the universe like symmetry violation.
Zhendong Zhang (Ph.D. now at Stanford University) and Kai-Xuan Yao (Ph.D. now at Citadel) were also co-authors on the paper.
More information:
Zhendong Zhang et al, Many-body chemical reactions in a quantum degenerate gas, Nature Physics (2023). DOI: 10.1038/s41567-023-02139-8
Provided by
University of Chicago
Citation:
Scientists observe first evidence of ‘quantum superchemistry’ in the laboratory (2023, August 7)