Scientists have achieved a series of milestones in growing a high-quality thin film conductor, suggesting in a new study that the material is a promising candidate platform for future wearable electronics and other miniature applications.
Researchers at The Ohio State University, the Army Research Laboratory and MIT determined that the material is the best among similarly built films for its electron mobility—an index of how easy it is for an electrical current to pass through it.
Coupled with low defect density to reduce interference with electron movement on the surface, the material is like a tiny empty freeway where all the electrons can easily get where they need to go with no traffic to be seen.
“We redefined what a car on this highway does—it’s like a car that can go really fast without getting encumbered by other things on the road,” said first study author Patrick Taylor, a physicist at the Army Research Laboratory.
“Future generations of electronics will use that kind of technology because it’s low-power,” Taylor said. “The Army is interested in low power because they don’t want to give a soldier something that hogs their battery. On the flip side, the commercial sector is looking at this kind of technology for what happens after silicon, because silicon’s reaching the end of its road and there has to be something that follows it.”
The research team reported the findings in Materials Today Physics.
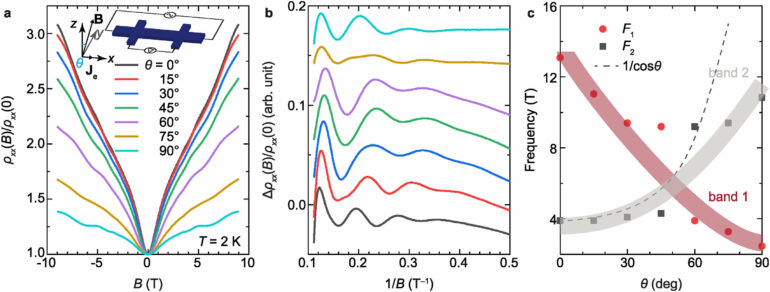
Co-lead author Brandi Wooten, a recent Ph.D. graduate in materials science and engineering at Ohio State who is now a research technician in mechanical and aerospace engineering, noted that exhaustive testing of the materials produced another milestone: Researchers were able to detect elusive oscillations that confirmed the pristine films were nearly scatter-free—unlike their counterparts in nature.
“These materials, naturally speaking, just aren’t the best quality in terms of thin film growth, but we need thin films to make devices,” Wooten said. “This is a nice paper showing we can make these materials good enough in thin film form to be put into devices. This is a steppingstone to getting these materials to do more.”
Part of doing more would likely involve taking advantage of—and expanding upon—the films’ thermoelectric capabilities. Wooten, who interned at Taylor’s lab for two summers while pursuing her Ph.D., oversaw highly sensitive tests to gauge the thin films’ thermal properties for this study, and the team has already begun working on new versions of the films based on what she found.
Though military and commercial applications are years away, these films, consuming very little energy, could be integrated with the super-thin chips now fabricated for miniature electronics. Potential uses could include serving as a basic building block for the next generation of magnetic memory in computers or to generate energy that powers robots or drones—or even wearable devices that keep soldiers cool while they’re wearing heavy gear and bulletproof vests.
The thin films—between 90 and 150 nanometers thick—are refined versions of ternary tetradymite, a mineral consisting of bismuth, tellurium and sulfur. For about two decades, scientists have focused on perfecting tetradymite films because of their potential to function as topological insulators: materials in which electrical current flows on the surface while the interior acts as an insulator, reducing any dissipation of the surface flow. This surface conduction also has spin properties, which could open the door to spintronic devices that use very low levels of power.
To achieve those properties, Taylor built the thin films using a technique called molecular beam epitaxy (MBE)—starting with the same crystal structure as tetradymite, but substituting other elements to come up with two different compositions that feature separate conduction mechanisms.
Joseph Heremans, a co-lead author of the paper, helped guide the selection of elements to arrive at the best films. A professor of mechanical and aerospace engineering, materials science and engineering, and physics at Ohio State, Heremans advised Taylor to aim for equilibrium while composing the materials—not a typical characteristic of films made with the MBE process.
“That was his guiding light,” Taylor said. “We did try to target more equilibrium conditions, and it paid off—and so the material we have has unusually high mobility.”
The high electron mobility is enabled by growing films in a way that reduces the concentration of moving particles carrying an electric charge that exist in the interior of natural tetradymites, Wooten said.
“By lowering that carrier concentration, we can utilize these really strong and robust states on the surface,” she said. “In topological insulators, the current can go in one direction on the surface, but not the other. It can’t back-scatter, and that’s what makes them more robust.”
This work represented an advance in being able to not just build these films, but to test their properties in the lab—previously, materials made for lab study were much larger.
“Using this molecular beam epitaxy technique, we can now envision a pathway toward something that might fit in your computer or cell phone someday,” Taylor said.
More information:
Patrick J. Taylor et al, Magnetotransport properties of ternary tetradymite films with high mobility, Materials Today Physics (2024). DOI: 10.1016/j.mtphys.2024.101486
Provided by
The Ohio State University
Citation:
A new material for small electronics that gives batteries longer life (2024, July 18)