From the tropics to the poles, from the sea surface to hundreds of feet below, the world’s oceans are teeming with one of the tiniest of organisms: a type of bacteria called Prochlorococcus, which despite their minute size are collectively responsible for a sizable portion of the oceans’ oxygen production. But the remarkable ability of these diminutive organisms to diversify and adapt to such profoundly different environments has remained something of a mystery.
Now, new research reveals that these tiny bacteria exchange genetic information with one another, even when widely separated, by a previously undocumented mechanism. This enables them to transmit whole blocks of genes, such as those conferring the ability to metabolize a particular kind of nutrient or to defend themselves from viruses, even in regions where their population in the water is relatively sparse.
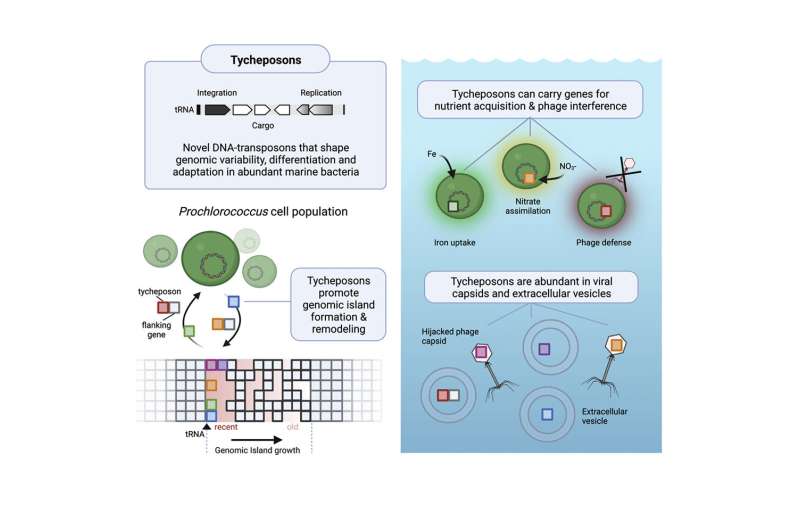
The findings describe a new class of genetic agents involved in horizontal gene transfer, in which genetic information is passed directly between organisms—whether of the same or different species—through means other than lineal descent. The researchers have dubbed the agents that carry out this transfer “tycheposons,” which are sequences of DNA that can include several entire genes as well as surrounding sequences, and can spontaneously separate out from the surrounding DNA. Then, they can be transported to other organisms by one or another possible carrier system including tiny bubbles known as vesicles that cells can produce from their own membranes.
The research, which included studying hundreds of Prochlorococcus genomes from different ecosystems around the world, as well as lab-grown samples of different variants, and even evolutionary processes carried out and observed in the lab, is reported today in the journal Cell, in a paper by former MIT postdocs Thomas Hackl and Raphaël Laurenceau, visiting postdoc Markus Ankenbrand, Institute Professor Sallie “Penny” Chisholm, and 16 others at MIT and other institutions.
Chisholm, who played a role in the discovery of these ubiquitous organisms in 1988, says of the new findings, “We’re very excited about it because it’s a new horizontal gene-transfer agent for bacteria, and it explains a lot of the patterns that we see in Prochlorococcus in the wild, the incredible diversity.” Now thought to be the world’s most abundant photosynthetic organism, the tiny variants of what are known as cyanobacteria are also the smallest of all photosynthesizers.
Hackl, who is now at the University of Groningen in the Netherlands, says the work began by studying the 623 reported genome sequences of different species of Prochlorococcus from different regions, trying to figure out how they were able to so readily lose or gain particular functions despite their apparent lack of any of the known systems that promote/boost horizontal gene transfer, such as plasmids or viruses known as prophages.
What Hackl, Laurenceau, and Ankenbrand investigated were “islands” of genetic material that seemed to be hotspots of variability and often contained genes that were associated with known key survival processes such as the ability to assimilate essential and often limiting nutrients such as iron, or nitrogen, or phosphates. These islands contained genes that varied enormously between different species, but they always occurred in the same parts of the genome and sometimes were nearly identical even in widely different species—a strong indicator of horizontal transfer.
But the genomes showed none of the usual features associated with what are known as mobile genetic elements, so initially this remained a puzzle. It gradually became apparent that this system of gene transfer and diversification was different from any of the several other mechanisms that have been observed in other organisms, including in humans.
Novel integrative elements and genomic plasticity in ocean ecosystems. © Cell (2023). DOI: 10.1016/j.cell.2022.12.006
Hackl describes what they found as being something like a genetic LEGO set, with chunks of DNA bundled together in ways that could almost instantly confer the ability to adapt to a particular environment. For example, a species limited by the availability of particular nutrients could acquire genes necessary to enhance the uptake of that nutrient.
The microbes appear to use a variety of mechanisms to transport these tycheposons (a name derived from the name of the Greek goddess Tyche, daughter of Oceanus). One is the use of membrane vesicles, little bubbles pouched off from the surface of a bacterial cell and released with tycheposons inside it. Another is by “hijacking” virus or phage infections and allowing them to carry the tycheposons along with their own infectious particles, called capsids. These are efficient solutions, Hackl says, “because in the open ocean, these cells rarely have cell-to-cell contacts, so it’s difficult for them to exchange genetic information without a vehicle.”
And sure enough, when capsids or vesicles collected from the open ocean were studied, “they’re actually quite enriched” in these genetic elements, Hackl says. The packets of useful genetic coding are “actually swimming around in these extracellular particles and potentially being able to be taken up by other cells.”
Chisholm says that “in the world of genomics, there’s a lot of different types of these elements”—sequences of DNA that are capable of being transferred from one genome to another. However, “this is a new type,” she says. Hackl adds that “it’s a distinct family of mobile genetic elements. It has similarities to others, but no really tight connections to any of them.”
While this study was specific to Prochlorococcus, Hackl says the team believes the phenomenon may be more generalized. They have already found similar genetic elements in other, unrelated marine bacteria, but have not yet analyzed these samples in detail. “Analogous elements have been described in other bacteria, and we now think that they may function similarly,” he says.
“It’s kind of a plug-and-play mechanism, where you can have pieces that you can play around with and make all these different combinations,” he says. “And with the enormous population size of Prochlorococcus, it can play around a lot, and try a lot of different combinations.”
Nathan Ahlgren, an assistant professor of biology at Clark University who was not associated with this research, says, “The discovery of tycheposons is important and exciting because it provides a new mechanistic understanding of how Prochlorococcus are able to swap in and out new genes, and thus ecologically important traits. Tycheposons provide a new mechanistic explanation for how it’s done.” He says, “they took a creative way to fish out and characterize these new genetic elements ‘hiding’ in the genomes of Prochlorococcus.”
He adds that genomic islands, the portions of the genome where these tycheposons were found, “are found in many bacteria, not just marine bacteria, so future work on tycheposons has wider implications for our understanding of the evolution of bacterial genomes.”
The team included researchers at MIT’s Department of Civil and Environmental Engineering, the University of Würzburg in Germany, the University of Hawaii at Manoa, Ohio State University, Oxford Nanopore Technologies in California, Bigelow Laboratory for Ocean Sciences in Maine, and Wellesley College.
More information:
Thomas Hackl et al, Novel integrative elements and genomic plasticity in ocean ecosystems, Cell (2023). DOI: 10.1016/j.cell.2022.12.006
Provided by
Massachusetts Institute of Technology
Citation:
Scientists discover a new way of sharing genetic information in a common ocean microbe (2023, January 5)



