Multiple sclerosis is characterized by an immune system gone haywire. A patient’s immune system starts treating the protective coating of the nerves – called myelin – as dangerous. The subsequent nerve damage can cause a variety of symptoms, including muscle weakness, pain and vision loss. MS currently has no cure, and doctors still don’t completely understand what causes it.
While there is a genetic component to MS, environmental factors also play a big role in determining whether someone will develop the disease. Recent evidence suggests that what’s in your digestive tract may also be a meaningful contributor to disease risk.
My colleagues and I at the University of Virginia are working to understand the two-way communication between the human body and the bacteria that live in its digestive system. In our recently published research, we found that bile acid in the intestines could be harnessed to protect people at high risk of MS from developing the disease, offering a new avenue for drug development.
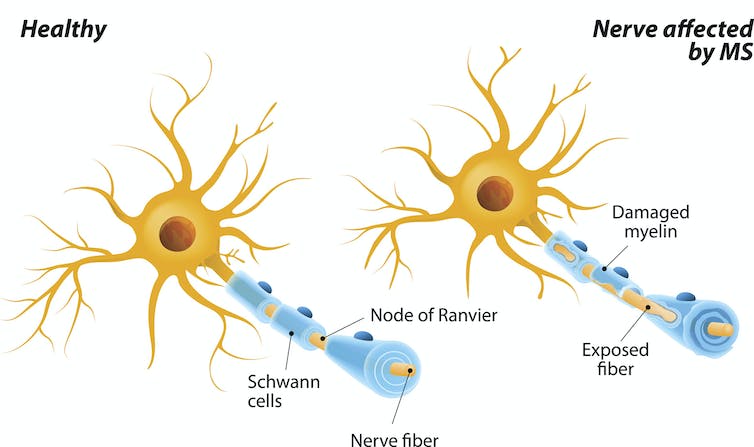
Multiple sclerosis results when the immune system attacks the protective myelin sheath of neurons.
ttsz/iStock via Getty Images Plus
The gut microbiome and autoimmunity
Trillions of bacteria live in the human gut. They help the body with everything from digesting food to preventing the overgrowth of infectious and dangerous bacteria. They also “educate” the immune system to recognize what is dangerous and what is not. If this process is disturbed, the immune system may become overactive and start to treat natural parts of the body as dangerous. This is called autoimmunity.
Scientists believe that one way bacteria and the immune system communicate with each other is through the aryl hydrocarbon receptor, or AHR, which resides in most cells of the body. This protein acts like an emergency call center – when it encounters certain chemicals, it will identify the appropriate response and send a signal to the cell recommending what it should do.
While researchers have shown that signals from AHR influence multiple sclerosis development, how it does so is unclear. To better understand what AHR is doing specifically in the guts of patients with MS, we genetically engineered mice that are missing AHR in some of their immune cells. By silencing AHR’s activity, we could understand what role it may be playing in autoimmunity.
We expected to learn more from this experiment about the molecular communication of immune cells. Instead we found something surprising: The gut environment in these mice had changed. Specifically, the chemical composition of their guts had been altered, indicating that the metabolism of gut bacteria had shifted. This meant that AHR is not only sensing what’s going on in the gut, but the receptor is also actively shaping its environment.