Like thieves that constantly look for ways to evade capture, Salmonella enterica, a disease-causing bacterium, uses various tactics to escape the human body’s defense mechanisms. In a new study, researchers from the Department of Microbiology and Cell Biology (MCB), IISc, highlight two such strategies that the bacterium uses to protect itself, both driven by the same protein.
When Salmonella enters the human body, each bacterial cell resides within a bubble-like structure known as Salmonella-containing vacuole (SCV). In response to the bacterial infection, the immune cells in our body produce reactive oxygen species (ROS) and reactive nitrogen species (RNS), along with pathways triggered to break down these SCVs and fuse them with cellular bodies called lysosomes or autophagosomes, which destroy the bacteria.
However, these bacteria have developed robust mechanisms to maintain vacuolar integrity, which is crucial for their survival. For example, when a bacterial cell divides, the vacuole surrounding it also divides, enabling every new bacterial cell to be ensconced in a vacuole. This also ensures that more vacuoles are present than the number of lysosomes which can digest them.
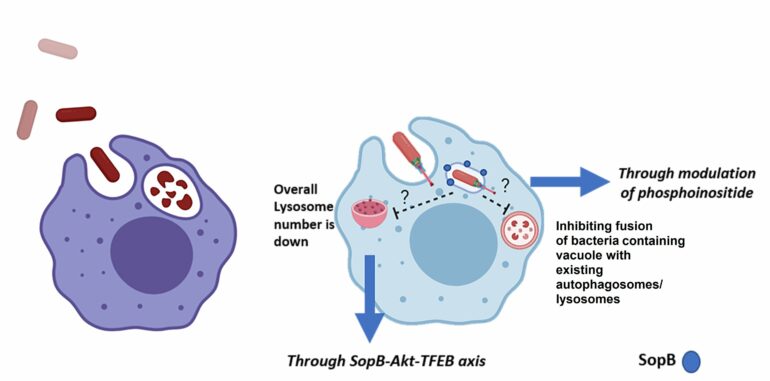
In the study published in Microbes and Infection, the IISc team deduced that a critical protein produced by Salmonella, known as SopB, prevents both the fusion of SCV with lysosomes as well as the production of lysosomes, in a two-pronged approach to protect the bacterium. “This gives the upper hand to bacteria to survive inside macrophages or other host cells,” explains Ritika Chatterjee, former Ph.D. student in MCB and first author of the study. The experiments were carried out on immune cell lines and immune cells extracted from mice models.
SopB acts as a phosphatase—it aids in removing phosphate groups from phosphoinositide, a type of membrane lipid. SopB helps Salmonella change the dynamics of the vacuole—specifically it alters the type of inositol phosphates in the vacuole membrane—which prevents the vacuole’s fusion with lysosomes.
A previous study from the same team had reported that the number of lysosomes produced by the host cells decreases upon infection with Salmonella. The researchers also found that mutant bacteria that were unable to produce SopB were also unable to reduce host lysosome numbers. Therefore, they decided to look more closely at the role that SopB was playing in the production of lysosomes, using advanced imaging techniques.
What they found was that SopB prevents the translocation of a critical molecule called Transcription Factor EB (TFEB) from the cytoplasm of the host cell into the nucleus. This translocation is vital because TFEB acts as a master regulator of lysosome production.
“This is the first time we deciphered that SopB can work in a dual manner—it changes the phosphoinositide dynamics of SCV and affects TFEB’s translocation into the nucleus. While other groups have already reported the function of SopB in mediating invasion in epithelial cells, the novelty of our study lies in identification of the function of SopB in inhibiting the vacuolar fusion with existing autophagosomes/lysosomes, and the second mechanism, which provides Salmonella with a survival advantage by increasing the ratio of SCV to lysosomes,” says Dipshikha Chakravortty, professor at MCB and corresponding author of the study.
The researchers suggest that using small molecule inhibitors against SopB or activators of TFEB can help counter Salmonella infection.
In subsequent studies, the team plans to explore the role of another host protein called Syntaxin-17 whose levels also reduce during Salmonella infection. “How do the SCVs reduce the levels of Syntaxin-17? Do they exchange it with some other molecules, or do the bacteria degrade it? We plan to look into it next,” says Chakravortty.
More information:
Ritika Chatterjee et al, Deceiving The Big Eaters: Salmonella Typhimurium SopB subverts host cell Xenophagy in macrophages via dual mechanisms, Microbes and Infection (2023). DOI: 10.1016/j.micinf.2023.105128. www.sciencedirect.com/science/ … ii/S128645792300031X
Provided by
Indian Institute of Science
Citation:
Stealthy Salmonella escapes host’s defenses using two-pronged approach (2023, May 26)