COVID-19 vaccines have been a game-changer in the current pandemic. Several vaccine candidates have conferred a high degree of protection, with some reducing the number of symptomatic infections by over 95% in clinical trials. But what determines this extent of protection? The answer to this question would help optimize the use of available vaccines and speed up the development of new ones.
Researchers at the Indian Institute of Science (IISc) and Queensland Brain Institute (QBI) in Australia have now addressed this question by developing a mathematical model that predicts how antibodies generated by COVID-19 vaccines confer protection against symptomatic infections. The study was published in Nature Computational Science.
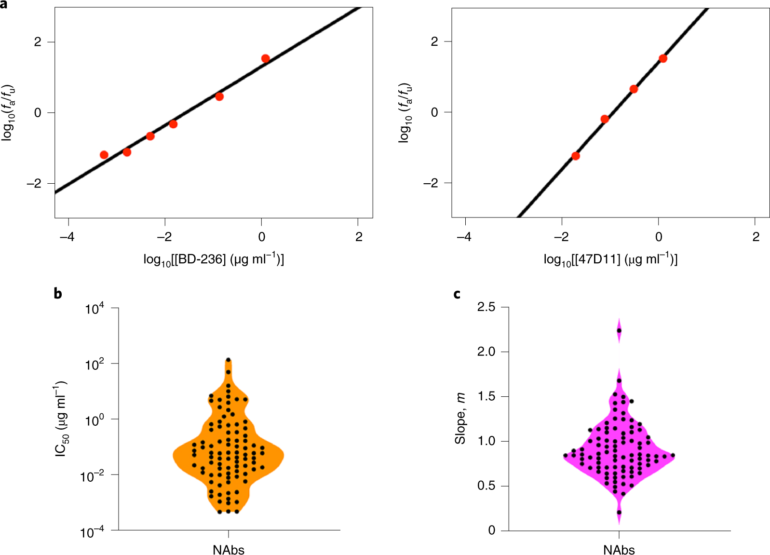
The researchers first analyzed over 80 neutralizing antibodies reported to be generated after vaccination against the surface spike protein of SARS-CoV-2, the virus that causes COVID-19. These antibodies are typically present in the blood for months and prevent virus entry by blocking the spike protein. The researchers hypothesized that these 80 antibodies constitute a “landscape” or “shape space,” and each individual produces a unique “profile” of antibodies that is a small, random subset of this landscape.
The team then developed a mathematical model to simulate infections in a virtual patient population of about 3,500 people with different antibody profiles, and to predict how many of them would be protected from symptomatic infection following vaccination.
“The reason predicting vaccine efficacies has been hard is that the processes involved are complex and operate at many interconnected levels,” says Narendra Dixit, professor at the Department of Chemical Engineering, IISc, and the senior author of the study. “Vaccines trigger a number of different antibodies, each affecting virus growth in the body differently. This in turn affects the dynamics of the infection and the severity of the associated symptoms. Further, different individuals generate different collections of antibodies and in different amounts.”
“This diversity of antibody responses was a challenge to comprehend and quantify,” adds Pranesh Padmanabhan, research fellow at QBI, the first author of the study.
The model developed by the team was able to predict the level of protection that would be conferred after vaccination based on the antibody “profile” of the individual, and the predictions were found to closely match efficacies reported in clinical trials for all the major approved vaccines.
The researchers also observed that vaccine efficacy was linked to a readily measurable metric called antibody neutralization titer. This opens up the possibility of using such models to test future vaccines for their efficacies before elaborate clinical trials are launched, the authors suggest.
Dixit, however, cautions that the study is based on current vaccines which have been designed to work on the original SARS-CoV-2 strain. “Our formalism is yet to be applied to the new variants, including Omicron, where other arms of the immune system and not just antibodies appear to be contributing to vaccine efficacies. Studies are ongoing to address this.”
More information:
Pranesh Padmanabhan et al, Modeling how antibody responses may determine the efficacy of COVID-19 vaccines, Nature Computational Science (2022). DOI: 10.1038/s43588-022-00198-0
Provided by
Indian Institute of Science
Citation:
A math model to predict COVID-19 vaccine efficacy (2022, March 1)