Rice University researchers have created an efficient, low-cost device that splits water to produce hydrogen fuel.
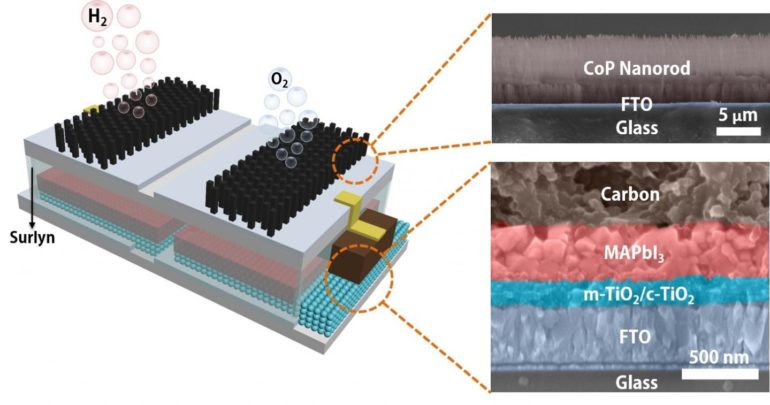
The platform developed by the Brown School of Engineering lab of Rice materials scientist Jun Lou integrates catalytic electrodes and perovskite solar cells that, when triggered by sunlight, produce electricity. The current flows to the catalysts that turn water into hydrogen and oxygen, with a sunlight-to-hydrogen efficiency as high as 6.7%.
This sort of catalysis isn’t new, but the lab packaged a perovskite layer and the electrodes into a single module that, when dropped into water and placed in sunlight, produces hydrogen with no further input.
The platform introduced by Lou, lead author and Rice postdoctoral fellow Jia Liang and their colleagues in the American Chemical Society journal ACS Nano is a self-sustaining producer of fuel that, they say, should be simple to produce in bulk.
“The concept is broadly similar to an artificial leaf,” Lou said. “What we have is an integrated module that turns sunlight into electricity that drives an electrochemical reaction. It utilizes water and sunlight to get chemical fuels.”
Perovskites are crystals with cubelike lattices that are known to harvest light. The most efficient perovskite solar cells produced so far achieve an efficiency above 25%, but the materials are expensive and tend to be stressed by light, humidity and heat.
“Jia has replaced the more expensive components, like platinum, in perovskite solar cells with alternatives like carbon,” Lou said. “That lowers the entry…